PROJECT HIGHLIGHTS
Governance Organization
MSU – Mount Sinai Hospital – University Health Network Academic Medical Organization
Project Title
Reusable Face Masks
Project Highlights
The ROSE Project team has successfully developed a design for a reusable, medical grade face mask that meets ASTM F2100-20 (Standard Specification for Performance of Materials Used in Medical Face Masks) Level 1 requirements for Sub-micron particle filtration efficiency at 0.1 micron (%) and Differential Pressure. To our knowledge, this goal has not been reached by any other reusable face mask in North America. Although there are 3 additional ASTM Level I Barrier Criteria to be met, as shown in Table 1 below, the particle filtration and differential pressure standards were the most challenging and essential standards that had to be met first (WHO guidance highlights three essential parameters: filtration, breathability, and fit). Testing is now underway for the remaining three standards at the Cambridge Materials Testing Lab. Based on previous testing of a four layer mask, we anticipate our final eight layer prototype will pass the remaining standards without difficulty or delay.
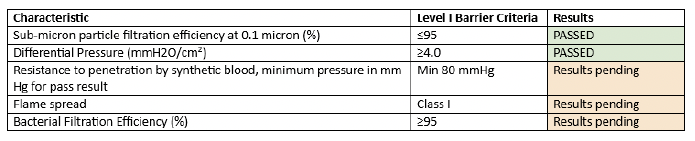
The ROSE mask design consists of eight layers of nonwoven polypropylene (NWPP) fabric (83g/m2) for it’s high filtration efficiency and fluid resistance, and one layer of woven cotton fabric as the inner layer for comfort. We completed testing on 20 different materials (silk, flannel, jersey, interfacing, NWPP, muslin, high TPI cotton, felt, polyester, and geotextiles) for particle filtration efficiency (PFE) and differential pressure (ΔP). These were split into two categories: filter medium and comfort layer. We then calculated a quality (Q) factor for each material (essentially high PFE and low ΔP), and continued our testing layering combinations of the materials. In total we have tested 48 different combinations of materials.
The NWPP materials had the highest Q factor. We sourced NWPP from three companies: OLYFUN, Smartfab and Uline. These materials were chosen as the filtration medium. The criteria for choosing the comfort layer was simply which woven cotton material had the lowest differential pressure. Muslin was chosen. After the many iterations of combining and layering these materials, we found that 8x Uline NWPP and 1x Muslin passed the criteria for ASTM Level 1 medical face mask with 96% PFE and 3.75 ΔP! Passing these standards is a major milestone for the project, after 20 months of research and testing. The ROSE mask is the only reusable medical face mask design in the world using readily available materials that anyone can purchase and fabricate. We are very excited to begin the next phase of the project. We will begin fit testing, washing and reuse testing, and mask efficacy research in the new year.
Theme
Methods of Risk Reduction and Mitigation
Primary Project Lead for Contact
Denyse Richardson, MD, MEd, FRCPC
Secondary Project Lead for Contact
Reena Kilian, MD, CCFP;Rachel Tyli, PhD (cand.)
